A booklet version of this publication is also available. You may print and distribute this freely provided that it is printed without alteration.
The birth control pill is currently being used by more than 10 million women in the US.1 A number of physicians and researchers have claimed that the birth control pill (BCP) (also called an oral contraceptive) is actually an abortifacient. An abortifacient is a substance that causes an abortion, the death of the zygote, embryo or fetus after conception has occurred. Others do not believe the BCP is an abortifacient as noted in a 1998 publication authored by several physicians: Hormonal Contraceptives: Are they Abortifacients? 2
This booklet uses an easy-reading question and answer format to review the available evidence for determining how the BCP works. All of that evidence, whether microscopic, macroscopic, or immunological, indicates that the BCP sometimes causes an early abortion. When and how often this happens remain unknown.
Questions Regarding the Birth Control Pill
Q1 What is a birth control pill (BCP) and how does it work?
Normally, as shown in Diagram A, the pituitary gland produces two hormones called FSH (Follicle Stimulating Hormone) and LH (Luteinizing Hormone). These hormones serve to stimulate the ovary to produce an egg each month (to ovulate). The ovary is also the production site for the woman’s two central female hormones: estradiol (EST), a type of estrogen, and progesterone (PRO), a type of progestin. BCPs are a combination of synthetic estrogen and progestin. BCPs “fool” the pituitary gland so that it produces less FSH and LH. By reducing the FSH and LH required for ovulation, BCPs suppress, but do not eliminate ovulation.

Birth control pills are acknowledged to have two other main effects:
- They thin the inner lining of the uterus (called the endometrium), depleting it of glycogen (a type of sugar) and blood supply, and
- BCPs may thicken the cervical mucus, making it more difficult for the sperm to travel up through the cervix.
Though this latter effect is claimed by BCP manufacturers, the evidence for it is weak4, 5 and not strongly supported by the rabbit model.6
Of course, BCPs could not cause abortions if they always stopped ovulation so this needs to be the first issue that is raised. A clear indication that ovulation will occur in women taking the BCP is provided by noting what the BCP manufacturers state in the PDR (Physician’s Desk Reference, 1998).7 The “efficacy rate” table for each BCP claims a “typical failure rate” of about 3%. The PDR defines “typical failure rate” as the rate of annual pregnancy occurrence in “typical couples who initiate use of a method (not necessarily for the first time) and who use it consistently and correctly during the first year if they do not stop for any other reason.” This means that even couples who use the pill consistently over the course of a year will have an average pregnancy rate of 3% according to the BCP manufacturers, who might tend to underestimate this number. A 1996 paper by Potter8 gives an excellent overview of the matter. She notes that the most recent data point to a rate of pregnancy for “typical use” as being 7%, which is probably the more accurate statistic, given the immediacy of her research data and the fact that today’s BCPs are lower dose and theoretically permit a higher rate of breakthrough ovulation. From these estimates of BCP failure and the common experience of on-pill pregnancies, it is clear that both ovulation and conception occur in couples who use the BCP.
Q2 Could you present the evidence that some physicians and researchers give for their claim that the pill is an abortifacient?
Before presenting that evidence, normal anatomy and histology (the study of the body’s tissues on a microscopic level) of the inner lining of the uterus (the endometrium) needs to be explained (see Diagram B).
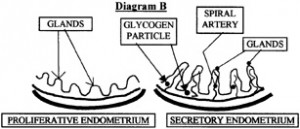
The endometrium slowly builds up before ovulation (the proliferative phase) and then peaks in the secretory phase (shortly after ovulation and possible conception). The endometrium is thus “ready for the newly conceived child to implant” a few days after ovulation. The blood flow carrying oxygen and nutrients to the glandular cells of the endometrium increases through the cycle as the spiral arteries enlarge during the secretory phase. The size of the endometrial glands also enlarges in the secretory phase. These glands contain important nutritional building blocks for the preborn child about to implant, including glycogen (a type of sugar), mucopolysaccharides (they supply certain building blocks for a cell’s growth) and lipids (fats).9
Q3 What does the phrase “ready for implantation” mean?
The author of a histology text designed for medical students states: “Thus, the various changes that take place in the endometrium during the second half of the menstrual cycle may be regarded as preparing the uterine lining for the nourishment and reception of the fertilized ovum (blastocyst).”10 It would appear that a woman’s body and the lining of her uterus are “optimal for implantation” a few days after ovulation and conception have occurred.
Q4 Does the BCP cause changes in the lining of the uterus that could be detrimental to the newly conceived child’s ability to implant himself or herself?
It would appear so. Since we know that the birth control pill sometimes allows ovulation and conception to occur, unfavorable changes in the endometrium could make it difficult for the preborn child to implant and would support the conclusion that the BCP is an abortifacient.
Q5 What are some of those changes?
The first change that the BCP makes is to markedly decrease the thickness of a woman’s endometrial lining. Women who take the pill know this because they can tell you that the volume of menstrual contents (flow) lost in their monthly cycles significantly decreases once they start taking the pill. Obviously, if a woman is losing less menstrual contents each month, the layer of endometrium that is being shed must be thinner and less well developed.
Q6 Is there a technical or quantitative way to measure how much thinner a woman’s endometrium becomes when she uses BCPs?
Yes, in 1991 researchers in the US performed MRI scans (Magnetic Resonance Imaging) on the uteri of women, some of whom were taking BCPs and some of whom were not.11 The BCP users had endometrial linings that were almost two millimeters thinner than those of the non-pill users. Although this may sound like a small difference, it represents a 57% reduction in endometrial lining thickness for women using BCPs.
Q7 But is there really any evidence that a thinner endometrium makes it more difficult for implantation to occur?
Yes. Several research papers have studied this issue, and it has been widely described in the medical literature concerning in-vitro fertilization where it has been noted that the newly conceived child is much less likely to implant in a thin uterine lining than a thick one. A small, older study [Fleisher et al12, 1985] did not find the thickness of the endometrium playing an important role in in-vitro implantation rates. However, later studies found a positive trend [Rabinowitz et al13, 1986; Ueno et al14, 1991] or a statistically significant effect [Glissant et al15, 1985] of decreasing endometrium thickness, thereby reducing the likelihood of implantation. Additional larger and more recent studies [Abdalla et al16, 1994; Dickey et al17, 1993; Gonen et al18, 1989; Schwartz et al19, 1997; and Shoham et al20, 1991] have reaffirmed this important conclusion. Most studies have found that a decrease of even one millimeter in thickness substantially decreases the rate of implantation. Two studies showed that when the endometrial lining became critically thin, no implantation occurred [Abdalla,16 Dickey17].
Q8 What does the actual endometrial-lining look like under a microscope for women who take BCPs ?

As shown in Diagram B, the uterine lining is at an “optimal state for implantation” when the glands and arteries are at their maximum size. This makes intuitive sense since, at this point, the blood supply and glycogen and lipid levels that the preborn child needs to survive are at their maximal state. Researchers who study the histology of the endometrium find that the BCP causes a number of changes to the endometrium. First, the spiral arteries regress significantly to the point where they are much smaller and may even be difficult to find under a microscope.21-24 This is important, since an adequate blood supply is critical to the existence of the implanting preborn child. A loss of blood flow means a drastic curtailment in the food and oxygen supply needed for the child’s survival. The blood flow to the endometrium is so important that in 1996 Kupesic wrote directly about its relationship to a preborn child’s likelihood of implantation.25 She found that the blood flow through the spiral arteries peaks at day 16 to 18 of the menstrual cycle, and then stated: “It seems that endometrial perfusion presents more accurate noninvasive assay of uterine receptivity than uterine artery perfusion alone. Therefore, blood flow velocity waveform changes of spiral arteries may be used to predict implantation success rate to reveal unexplained infertility problems and to select patients for correction of endometrial perfusion abnormalities…”26 (emphasis added). In layman’s language, Kupesic is stating that the likelihood of implantation correlates with the blood flow through the spiral arteries.
Q9 In addition to the reduced blood supply from the spiral arteries, what other microscopic level changes to the endometrium are caused by BCPs?
The second prominent effect is that the endometrial glands become much smaller and the “mitotic rate” (rate of cell division) of the cells of the glands decreases.21-24 Obviously if the glands which supply the glycogen (sugar), mucopolysaccharides, or lipids (fats) are compromised, the preborn child who needs those nutrients will have a more difficult time implanting and/or surviving.
Q10 Many of the studies that examined the endometrial lining are older and were performed when the estrogen content of BCPs was much higher (100 micrograms or more). Would the same effect be occurring with the more recent BCPs?
Yes. First it should be mentioned that if you ask a woman who is taking lower dose BCPs about the amount of monthly menstrual contents that she loses, she will note that she loses significantly less after she started taking the BCP. Obviously if she is losing less menstrual contents, then she is shedding less because the lining of the uterus has become thinner. Even histologic studies for BCPs which contain 50 micrograms of estrogen (a medium dose) and 0.5 mg of a progestin (norgestrel) found that the spiral arteries and the endometrial glands “shrivel up.”22-23
Q11 Is there any new evidence that supports the argument that BCPs act by causing an early abortion?
Yes. In 1996 a researcher named Stephen Somkuti published an article concerning the endometrium and a group of molecules called “integrins.” 27Integrins are a group of adhesion molecules that have been implicated as playing an important role in fertilization and implantation. There are several different types of integrins, and it is believed that the endometrium is most receptive to implantation when it expresses certain types of integrins. Birth control pills change the type of integrins that the endometrial lining produces, theoretically making it more difficult for the preborn child to implant. In the words of Dr. Somkuti: “These alterations in epithelial and stromal integrin expression suggest that impaired uterine receptivity is one mechanism whereby BCPs exert their contraceptive action.”28
Q12 Has anyone proven that the BCP causes early abortions?
In order to prove if and how often women are having abortions while taking BCPs, one needs to be able to measure how often women become pregnant while taking them. But early pregnancy tests are currently not accurate enough to confirm pregnancy within the first week, although some researchers have been able to detect the hormonal changes in pregnancy as early as four days after conception.29, 30 Until a very early pregnancy test is developed or until researchers physically measure how many abortions are occurring in women who take BCPs, one cannot state with absolute certainty when and how often BCPs cause early abortion. The most accurate description of the current evidence is as follows:
All of the evidence, whether at a microscopic, a macroscopic, or an immunological level, strongly supports the argument that the BCP causes an early abortion at times. Until further studies are done, we should take heed and base our actions and pronouncements on the current evidence.
Larimore and Stanford’s comprehensive review article [see Archives in Family Medicine, 2/2000] is an excellent resource for those interested in reading more about the Pill and the evidence for its abortifacient action.
Q13 Recently a group of physicians, many of whom are experienced Ob/Gyns, wrote a booklet entitled “Hormonal Contraceptives: Are they Abortifacients?” In it they write: “The hormonal contraception is abortifacient theory is not established scientific fact. It is speculation…” Could you comment on why a group of physicians would hold this view and on the nature of their arguments?
An overview and rebuttal to the arguments cited in the booklet entitled “Hormonal Contraceptives: Are they Abortifacients?”2 are presented here as an Appendix. This author believes that some of their own arguments can be shown to actually support the position that the pill is an abortifacient.
Q14 How frequently do BCPs cause an early abortion?
At this point, no one knows. There are many factors which influence the answer to this question and it is possible that as technology improves, an accurate estimate will be made. One of the determining factors is how often BCPs allow ovulation to occur. If the rate of ovulation is documented to be substantially higher than the pregnancy rate, then one could start to make an estimate of the frequency of abortion in women who take the BCP.
But measuring a woman’s ability to ovulate is difficult. Researchers measure ovulation rates in women who are taking the pill by using several parameters including: 1) Ultrasound measurements of the ovary, specifically the size of the largest (dominant) follicle (which contains the egg or oocyte); and 2) hormonal assays of progesterone and estradiol levels. Until now, many researchers have arbitrarily accepted that a pregnancy has occurred when the progesterone levels reaches a certain level. But it is possible that BCPs depress the ovary’s ability to produce progesterone despite pregnancy, as noted as early as 1962 by Holmes et al.31 It would seem more accurate to measure ovulation rates based on daily pelvic or vaginal ultrasound exams. In 1985, Ritchie32 wrote in his review of the role of ultrasound in the evaluation of normal and induced ovulation: “With daily scanning, ovulation can be demonstrated in >80% of cases.” In a 1998 paper Petta et al33 commented on ultrasound in regard to ovulation: “Follicles that disappeared or that were abruptly reduced in size by > 50% after reaching a diameter of 15 mm were considered to have ruptured.”
There are a number of other reasons why determining the frequency of ovulation by such a method is important. First, studies of women who take the pill often show a high rate of “ovarian activity” in their dominant follicles which may reach a size that is consistent with those seen in non-BCP users who ovulate. In other words, the ultrasound measurements indicate that these women (the BCP users) are about to ovulate. But these same studies often conclude that ovulation has not occurred because the progesterone level has not reached a critical level.34, 35 This is somewhat counter-intuitive in light of a recent study35 that found: “Patients using the lower-dose monophasic and multiphasic pills had follicular activity similar to that of those using nonsteroidal contraception, with the important exception that ovulation rarely occurred.” This study, as almost all others, used the criteria that ovulation is confirmed when a progesterone levels reaches a certain level. This may not be accurate.
High-tech ultrasound may reveal that ovulation rates are higher than today’s commonly quoted rates of 3-5%.32 The two reasons for this are that today’s BCPs contain far less estrogen and progestin than the early BCPs did and therefore suppress ovarian activity less often. Second, many studies have examined the rate of breakthrough ovulation in women who have recently started taking the pill, but the question that must be asked is: “Does the rate of ovulation go up in women who have taken BCPs for more than a year?” This phenomenon occurs with Norplant, where it was noted that the breakthrough ovulation rate in the first year was only 11%, but increased dramatically after that year, so that a 7-year average yielded an annual breakthrough ovulation rate of 44%36(although part of the reason for this increase may have been declining Norplant hormone levels with time). But could a woman’s pituitary gland “compensate” or “reset itself” to adjust for the presence of the hormones in the BCP so that ovulation occurs more frequently with time? If so, future trials may show that the rate of breakthrough ovulation increases in women who take the low dose BCP for longer periods of time.
It seems likely that a study will be done in the future that measures the rate of ovulation based on serial ultrasounds (although some may claim that such a study might be unethical). If such a study is performed in women who have been taking low dose BCPs for longer than a year, it could yield information that leads to a more credible estimate of the abortion rate for women taking BCPs.
Questions Regarding other Contraceptives
Q15 Does the intrauterine device (IUD) cause abortion?
Yes, the IUD does not prevent ovulation37 and works by changing the inner lining of a woman’s uterus so that the newly conceived child cannot implant in the womb.
Q16 Do groups who favor abortion admit that BCPs and the IUD work by causing an early abortion?
The most ardent pro-abortion supporters openly admit the abortifacient nature of the BCP and the IUD. In his arguments before the Supreme Court in 1989, in the case of Webster versus Reproductive Health Services, Mr. Frank Susman, arguing for the pro-abortion side, spoke to Justice Anthony Scalia: “If I may suggest the reasons in response to your question, Justice Scalia. The most common forms of what we generally in common parlance call contraception today, IUD’s, and low-dose birth control pills, which are the safest type of birth control pills available, act as abortifacients. They are correctly labeled as both.”38
Q17 Do other hormonal “contraceptives” such as the long-acting progestins cause early abortions?
Norplant, manufactured by Wyeth-Ayerst, and Depo-Provera made by Pharmacia-Upjohn are made of artificial progestins. Norplant is composed of levonorgestrel and Depo-Provera of medroxyprogesterone. Depo-Provera is a long-acting progestin that is injected every three months intramuscularly. It is used worldwide despite the fact that animal and human studies have all shown that it increases the risk of breast cancer by at least 190% in women who took it for more than two years before the age of 25!39 Norplant is an artificial progestin that consists of a series of Silastic (rubber-like) strips which are filled with levonorgestrel and implanted under the skin of a woman’s upper arm and slowly release the progestin into the woman’s body over a five year time period. Norplant has been found to allow breakthrough ovulation in over 44% of a woman’s monthly cycles.36 In addition, a study of rabbits conducted by Chang40 has shown that sperm freely reached the rabbit’s fallopian tubes-even when the rabbits were given high doses of synthetic progestin. The combination of a high rate of breakthrough ovulation and documented sperm migration to the fallopian tubes in animals suggests that progestins such as Norplant and Depo-Provera allow a high rate of fertilization and subsequent abortion, most likely even higher than BCPs.
Q18 Does the “the morning after pill” cause an early abortion?
The “morning after pill” consists of a series of high dose BCPs that some women have taken one or two days after thinking that they have conceived. These high dose hormones act as an abortifacient by thinning the lining of the uterus, thus preventing the newly conceived child from implanting. The animal model described by Castro-Vazquez in 1971 demonstrated this effect in rats.41
Q19 Some emergency rooms give “hormones” to women who have recently been raped. Can this cause an early abortion?
The woman who comes to the emergency room within a few hours of being raped may or may not have already conceived. Some ER staff will give such a woman high dose estrogen and progestin hormones very similar to the “morning after pill.” The exception is often found in Catholic hospitals whose physicians may not give the “post-rape pill.” For the woman who is raped near the time of ovulation, the hormones may indeed stop ovulation and prevent conception. But if ovulation and conception have occurred, the hormones will work by causing an early abortion in the same way as has been described for “the morning after pill.” Since there is no way to know whether conception has occurred, some physicians will not prescribe the “post-rape pill.”
Q20 Does artificial fertilization cause early abortion(s).
Every method of artificial fertilization that this author is aware of, whether it be in vitro fertilization or ZIFT (zygote intrafallopian transfer) or GIFT (Gamete intrafallopian transfer), involves the death of many unborn children during the process. Fewer than one out of 20 conceived children “survive” the process of in vitro fertilization. Even GIFT involves the exposure of more than one egg to multiple sperm – a situation in which multiple early abortions are extremely likely to occur. In addition to these methods, it is possible that women who take fertility pills such as Clomid (which work by causing the ovaries to “super-ovulate”) may be experiencing early abortion(s). Some studies,42-45 but not all,46 indicate that this drug thins the lining of the uterus, theoretically making it more difficult for the conceived child(ren) to implant.
Q21 Can the estrogens that women take “after menopause” cause an early abortion?
Often women are started on estrogen replacement near the time of menopause. This usually has a beneficial effect of reducing the risk of osteoporosis, while increasing the risk of uterine and breast cancer. Unfortunately, many women are now starting estrogen replacement before they have completely stopped ovulating; that is, they are not always in true menopause but are still having occasional cycles. If a woman were to start estrogen at a time when she was having an occasional cycle, she could still conceive and have an early abortion. This is something to be aware of, and women who wish to avoid this effect should not start hormonal replacement therapy until they have not had a cycle for a one-year period.
Q22 Why was the term “contraceptive” placed in quotations when referring to the various artificial hormones?
Oral “contraceptives”, Norplant, Depo-Provera, the IUD, the “morning after pill,” and the “post-rape pill” all work by causing an early abortion at least part of the time. The word “contraceptive” was placed in quotations because all of the evidence points to these hormones or procedures as being abortifacients. That is, they cause an early abortion some of the time. Contraception technically means, “to prevent conception.” Clearly, the hormones alluded to here cause the death of the preborn child after conception and cannot accurately be solely called “contraceptives.”
Appendix
Response to the arguments put forth in the brochure titled: Hormonal Contraceptives: Are they Abortifacients?
In January 1998, a group of twenty-two physicians (almost all are Ob/Gyns) wrote a collaborative report questioning the abortifacient nature of the pill.2 Their four main arguments (taken from page 7 in their booklet) and a corresponding rebuttal to each are presented here
1: They write: “We know of no existing scientific studies that validate the ‘hormonal contraception is partly abortifacient’ theory. ‘On-pill’ pregnancy rates roughly parallel ‘on-pill’ ovulation rates (about 3-5 percent on the 35 mcg pill). Increased spontaneous abortion of on-pill pregnancies is not noted.”
Here the term “pregnancy rate” refers to the rate of pregnancy as determined by a positive pregnancy test. We must acknowledge that a woman is actually pregnant directly after conception, before one can diagnose it by a simple pregnancy test. The claim that “on-pill” pregnancy rates roughly parallel “on-pill” ovulation rates may appear to be a satisfying argument, but on closer examination this contention actually bolsters the argument in favor of the pill acting as an abortifacient. Why? If a woman is taking the pill, she will experience an artificially regulated cycle that lasts 28 days so she will have about 13 cycles per year (365 days divided by 28). Thus a group of 100 women would be expected to have a total of 1300 cycles per year. If women taking the pill experience a breakthrough ovulation rate (on-pill ovulation rate) of between 3% and 5%, a group of 100 women would be expected to have between 39 and 65 breakthrough ovulation cycles in one year (1300 x 3% – 5%). William’s Obstetrics notes that the average woman has a “natural fecundibility rate” of 28 percent.47 “Natural Fecundiblity rate,” perhaps more accurately called the fertility rate, is defined in this section of William’s Obstetrics asliveborn infants per ovarian cycle.
But William’s Obstetrics also notes that for every 600 liveborn children, 279 embryos or fetuses are miscarried, 176 of them after a positive pregnancy test and 103 of them prior to being able to detect that a woman is pregnant. This means that the average couple will actually have a detectable pregnancy rate of {28%+ (176/600 x 28%)}= 36.2%. So a group of 100 women who are sexually active and using the birth control pill might expect between 14 and 24 detectable pregnancies per year: [{39 – 65} x 36.2%]. But the PDR (Physician’s Desk Reference) notes that a group of 100 women who are using the pill in a consistent manner will have about 3 pregnancies per year7 and a recent study by Potter yields an updated statistic of 7 pregnancies per year.8 If it is true that “on-pill pregnancy rates roughly parallel on-pill ovulation rates,” then the conclusion that the pill is not an abortifacient is highly suspect. This is because if the ovulation rate is 3% to 5%, we might expect the pregnancy rate to be 14% to 24% __ that is, far higher than the ovulation rate. Since we do not see this clinically, we must ask: Why is the clinically measurable pregnancy rate far lower than the theoretical rate based on the rate of breakthrough ovulation?
A number of explanations exist including the failure of sperm to reach the egg due to thicker cervical mucus or a change in motility within the fallopian tubes which the pill may cause. But one must also recognize that the difference in rates may be due to a failure of the zygote/embryo to implant due to the pill’s effects on the endometrial lining. In short, the observation that “on-pill pregnancy rates roughly parallel on-pill ovulation rates” serves, if anything, to support the argument that the pill is an abortifacient.
2: They write: “There is regular successful implantation of the invasive blastocyst on surfaces a great deal more ‘hostile’ than ‘hostile endometrium’ (eg, fallopian tube lining). ‘Hostile endometrium’ is not a demonstrated clinical reality.”
This argument is specious. It has already been stated in the answers to questions 3-13 that the sum of the evidence, both recent and old, supports the argument that the pill changes the lining of the endometrium in a fashion unfavorable for implantation. The fact that the preborn child may attach him or herself to a structure such as the fallopian tube lining has little to do with the previous arguments. Although one can make the argument that a rare occurrence or an exception disproves a theory, one cannot deduce the converse, namely, that the exception proves the theory. That is, noting that some preborn children do implant in the fallopian tube, or for that matter in the peritoneal cavity, merely proves that it is possible for this event to occur. But it offers no evidence that justifies the claim that a favorable implantation site is just as good as an unfavorable one.
3: They write: “The extremely rare reporting of ectopic pregnancies associated with hormonal contraception would indicate the rarity of actual conception by patients using these modalities.”
Once again the noted physicians apparently were unaware that their statement serves the purpose of supporting the pill’s action as an abortifacient. It should be stated that both women who take the pill, and those who do not, can and do become pregnant. The pregnancy can be an extrauterine pregnancy (EUP) {usually a tubal pregnancy} or an intrauterine pregnancy (IUP) {the normal type of pregnancy}. One can measure the ratio of EUP to IUP in either group. What should happen to this EUP/IUP ratio if one compares women who are not taking the pill to those who are?
The Ob/Gyns would argue that this ratio should remain constant and if the reporting of ectopic pregnancy were “practically unreported,” as the Ob/Gyns write, one might even expect the ratio to decrease, since the numerator would become smaller. On the contrary, if the pill caused more early abortions (less intrauterine pregnancies), one would expect the number of intrauterine pregnancies (IUPs) to decrease in comparison to the number of extrauterine pregnancies (EUPs) and thus the ratio should increase. What does the literature say?
The studies to date show that women who take the pill have an increased ratio of EUP to IUP. They note that women who take the pill are far more likely to experience more EUPs per IUP than women who do not take the pill, which supports the argument that the pill is an abortifacient. The reported odds ratios ( an odds ratio of 2.0 is the same as saying a two-fold risk) of the increased risk of EUP/IUP in women taking the pill compared to women who were not taking the pill were as follows: 1) WHO48 found an odds ratio of 1.7 (1.1-2.5); 2) Mol et al49 found an odds ratio of 1.8 (0.9-3.4); 3) Job-Spira et al50 found an odds ratio of 4.3 (1.5-12.6); 4) Thorburn et al51 found an odds ratio of 4.5 (2.1-9.6); and 5) Coste et al52 found an odds ratio of 13.9 (1.8-108.3). These clinical studies once again provide evidence that suggests that the pill acts as an abortifacient.
4: They write: “Many factors play a part in how a family plans and spaces their children. It is not the purpose of this paper to promote nor to oppose hormonal contraception.”
As a physician I know that it is common to use a medicine or a type of procedure because previous physicians have done so. It is simply impossible for each physician to “re-invent the wheel” when trying to decide if a particular drug or procedure is the optimal one. Unfortunately, once one becomes accustomed to particular ways of doing things, one tends to continue to do them in a particular fashion because “they have always been done that way,” and “new thoughts” on a “standard procedure” are not always appreciated.
How do these statements pertain to the current argument? It has been stated that almost every physician who signed or helped write the booklet Hormonal Contraceptives: Are they Abortifacients? is or was an obstetrician/gynecologist. It is common knowledge that virtually all Ob/Gyns prescribe the pill to their patients for contraception, in addition to other indications. Therefore, I assume (and would certainly issue a retraction were I proven wrong) that nearly every Ob/Gyn who signed or helped write the paper currently prescribes or has prescribed birth control pills for contraception.
The problem here is that self-proclaimed pro-life Ob/Gyns would have difficulty being unbiased in any argument about whether the pill causes early abortions, since each of these physicians most likely has written thousands of oral contraceptive prescriptions in their careers. If they were to admit that the pill is an abortifacient, they would be admitting that they had likely aborted hundreds of tiny preborn children. Surely it would be difficult for a pro-life obstetrician to fairly evaluate the abortifacient action of the pill given these circumstances.
5: Recently Decook et al53 have argued that if a breakthrough cycle does occur while a woman is taking the pill, her endometrial lining would become similar to that of the non- BCP user for that cycle. Is this an accurate statement?
To the best of this author’s knowledge, that statement has no support in the medical literature. If the above statement were true, it would mean that each time a woman had a breakthrough cycle while taking the BCP (if she does not become pregnant), she should experience as heavy a cycle as if she were not taking the pill. This phenomenon has not been described in the medical literature either.
Conclusion: The arguments presented by the twenty-two physicians in the booklet entitled Hormonal Contraceptives: Are they Abortifacients? lack substance and actually serve to bolster the evidence that the birth control pill causes early abortions.
Footnotes
1. Faust JM. Image change for condoms. ABC News Report. [Internet E-mail]. 6/8/97.
2. DeCook JL, McIlhaney J et al. Hormonal Contraceptives: Are they Abortifacients: 1998; Frontlines publishing. Sparta, MI. For contact information call 1-616-887-6256. Email: order@frontlines.org
3. Smith, Janet. Contraception, Why Not? One More Soul. Dayton, OH (1-937-279-5433)
4. Elstein M, et al. Studies on low dose oral contraceptives: cervical and plasma hormone changes in relation to circulating d-norgestrel and 17alpha-ethyniyl estradiol concentrations. Fertility and Sterility. 27; 1976: 892-899.
5. Wolf DP, et al. Human cervical mucus v. oral contraceptives and mucus rheologic properties. Fertility and Sterility. 32; 1979: 166-169.
6. Chang MC, Hunt DM. Effects of various progestins and estrogen on the gamete transport and fertilization in the rabbit. Fertility and Sterility. 1970; 21: 683-686.
7. Physicians’ Desk Reference: 1997 {The noted information can be found when looking up any oral contraceptive. Failure rate for “typical use” is noted to be 3 percent.}
8. Potter LA. How effective are contraceptives? The determination and measurement of pregnancy rates. Obstet Gynecol. 1996; 88: 13S-23S.
9. Snell, Richard. Clinical and Functional Histology for the Medical Student. Little, Brown & Co. Boston; 1984, 586-591.
10. Ibid.
11. Brown HK, et al. Uterine Junctional Zone: Correlation between Histologic Findings and MR Imaging. Radiology. 1991; 1798: 409-413.
12. Fleischer AC et al, Sonography of the endometrium during conception and nonception cycles of in vitro fertilization and embryo transfer. Fertility and Sterility. 1986; 46: 442-447.
13. Rabinowitz R, et al. The value of ultrasonographic endometrial measurement in the prediction of pregnancy following in vitro fertilization. Fertility and Sterility. 1986; 45: 824-826.
14. Ueno J, et al. Ultrasonographic appearance of the endometrium in natural and stimulated in vitro fertilization cycles and its correlation with outcome. Human Reproduction. 1991; 6: 901-904.
15. Glissant A, et al. Ultrasound study of the endometrium during in vitro fertilization cycles. Fertility and Sterility. 1985. 44: 786-789.
16. Abdalla HI, et al. Endometrial thickness: a predictor of implantation in ovum recipients? Human Reproduction. 1994; 9: 363-365.
17. Dickey RP, et al. Relationship of endometrial thickness and pattern to fecundity in ovulation induction cycles: effect of clomiphene citrate along and with human menopausal gonadotropin. Fertility and Sterility. 1993. 59: 756-760.
18. Gonen Y, et al. Endometrial thickness and growth during ovarian stimulation: a possible predictor of implantation in in vitro fertilization. Fertility and Sterility. 1989; 52: 446-450.
19. Schwartz LB, et al. The embryo versus endometrium controversy revisited as it relates to predicting pregnancy outcome in in vitro fertilization embryo transfer cycles. Human Reproduction. 1997; 12: 45-50.
20. Shoham Z, et al. Is it possible to run a successful ovulation induction program based solely on ultrasound monitoring: The importance of endometrial measurements. Fertility and Sterility. 1991; 56: 836-841.
21. Hilliard George D, Norris HJ, Pathologic Effects of Oral Contraceptives, Recent Results in Cancer Research. 1979. 66;49-71.
22. Ober WB. The effects of oral and intrauterine administration of contraceptives on the uterus. Human Pathology. 1977; 8: 513-527.
23. Ober WB. Synthetic progestagen-oestrogen preparations and endometrial morphology. J. Clin Path. 1966; 19: 138.
24. Roland M, et al. Sequential endometrial alterations during one cycle of treatment with synthetic progestagen-estrogen compounds. Fertility and Sterility. 1966. 17: 339.
25. Kupesic S. The first three weeks assessed by transvaginal color doppler. J. Perinat. Med. 1996; 24: 301-317.
26. Ibid.
27. Somkuti SG, et al. The effect of oral contraceptive pills on markers of endometrial receptivity. Fertility and Sterility. 1996. 65; 484-488.
28. Ibid.
29. Witt B, Wolf G, et al. Relaxin, CA-125, progesterone, estradiol, Schwnagerschaft protein, and human Chorionic Gonadotropin as predictors of outcome in threatened and nonthreatened pregnancies. Fertility and Sterility. 1990; 53: 1029-1036.
30. Norman RJ, et al. Inhibin and relaxin concentration in early singleton, multiple, and failing pregnancy: relationship to gonadotropin and steroid profiles. Fertility and Sterility. 1993; 59: 130-137.
31. Holmes, et al. Oral contraceptives: An assessment of their mode of action. The Lancet. June 2, 1962. 1174-1178.
32. Ritchie WGM. Ultrasound in the evaluation of normal and induced ovulation. Fertility and Sterility. 1985; 43: 167-181.
33. Petta CA, et al. Timing of onset of contraceptive effectiveness in Depo-Provera users. II. Effects on ovarian function. Fertility and Sterility; 70: 817-820.
34. Van der Vange N. Ovarian activity during low dose oral contraceptives. Contemporary Obstetrics and Gynecology. G. Chamberlain. London, Butterworths, 1988, 315-326.
35. Grimes DA, et al. Ovulation and follicular development associated with three low-dose oral contraceptives: A randomized controlled trial. Obstetrics & Gynecology. 1994; 83: 29-34.
36. Croxatto HB, Diaz S, et al. Plasma progesterone levels during long-term treatment with levonorgestrel silastic implants. Acta Endocrinologica. 1982; 101: 307-311.
37. Ibid.
38. Alderson Reporting Company. Transcripts of oral arguments before court on abortion case. New York Times. April 27, 1989; B12.
39. Skegg DCG, Noonan EA, et al. Depot medroxyprogesterone acetate and breast cancer [A pooled analysis of the World Health Organization and New Zealand studies]. 1995; JAMA: 799-804.
40. Chang MC, Hunt DM. Effects of various progestins and estrogen on the gamete transport and fertilization in the rabbit. Fertility and Sterility. 1970; 21: 683-686
41. Castro-Vazquez. Macome JC, et al. On the mechanism of action of oral contraceptives. Effect of Lynestrenol on ovum implantation and oviductal morphology in the rat. Fertility and Sterility. 1971; 22: 741-744.
42. Eden JA et al The effect of Clomiphene citrate on follicular phase increase in endometrial thickness and uterine volume. Obstet. Gyn. 1989; 73: 187-190.
43. Yagel S, et al. The effect of ethinyl estradiol on endometrial thickness and uterine volume during ovulation induction by clomiphene citrate. Fertility and Sterility. 1992. 57: 33-36.
44. Fleischer AC, et al. Sonographic depiction of endometrial changes occurring with ovulation induction. J of Ultrasound Med. 1984; 3: 341-346.
45. Imoedemhe DA, et al. Ultrasound measurement of endometrial thickness on different ovarian simulation regimens during in vitro fertilization. Hum Reprod. 1987; 2: 545-547.
46. Dickey RP, et al. Relationship of endometrial thickness and pattern to fecundity in ovulation induction cycles: effect of clomiphene citrate alone and with human menopausal gonadotropin. Fertility and Sterility. 1993. 59: 756-760.
47. Cunningham, et al. Williams Obstetrics, 20th Edition. Appleton and Lange. 1997. Stanford, CT. p 580-1.
48. The WHO Task Force on intrauterine devices for fertility regulation. A multinational case-control study of ectopic pregnancy. Clin Reprod Fertil 1985;3:131-143.
49. Mol BWJ, Ankum WM, Bossuyt PMM, and Van der Veen F. Contraception and the risk of ectopic pregnancy: a meta analysis. Contraception 1995; 52: 337-341.
50. Job Spira N, Fernandez H, Coste J, Papiernik E, Spira A. Risk of Chlamydia PID and oral contraceptives. J Am Med Assoc 1990;264:2072-4.
51. Thorburn J, Berntsson C, Philipson M, Lindbolm B. Background factors of ectopic pregnancy. I. Frequency distribution in a case-control study. Eur J Obstet Gynecol Reprod Biol 1986;23:321-331.
52. Coste J, Job-Spira N, Fernandez H, Papiernik E, Spira A. Risk factors for ectopic pregnancy: a case-control study in France, with special focus on infectious factors. Am J Epidemiol. 1991;133:839-49.
53. DeCook J, et al. Hormonal Contraceptives, Controversies and Clarification. February, 1999. Pro-Life Obstetrician. PO Box 81, Fennville, MI 49408






